This is an automatically translated article.
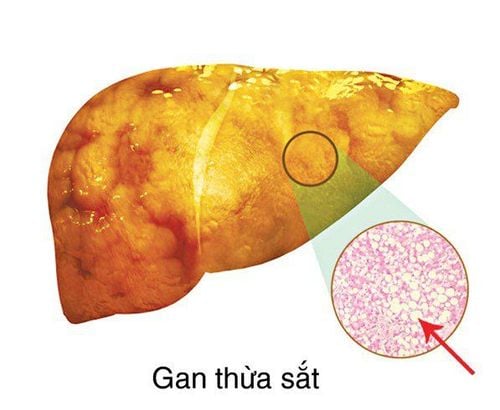
Defothal 125mg is a drug used in the treatment of chronic iron overload, suitable for patients with beta thalassemia major due to frequent blood transfusions, non-transfusion-dependent thalassemia syndrome and some other anemias. The use should be strictly according to the instructions and supervision from the doctor to achieve the highest effectiveness.
1. What is Defothal 125?
Defothal 125 contains the active ingredient Deferasirox 125mg. This is the active ingredient that helps remove excess iron from the body (also known as iron overload). The drug works by trapping and removing excess iron in the body, then the iron will be excreted mainly through feces.
2. Indications and contraindications to the drug Defothal 125
2.1. Indications Defothal 125 Defothal 125 is indicated for use in overcoming chronic iron overload due to frequent blood transfusions, which are common in patients with beta thalassemia major aged 6 years and older. Defothal 125 is also used in the treatment of transfusion-induced chronic iron overload when Deferoxamine therapy is contraindicated or inadequate in patient groups such as: Patients 2 to 5 years of age with severe beta thalassemia iron from frequent blood transfusions. Adults and children over 2 years of age with beta thalassemia major with iron overload due to infrequent blood transfusions. Adults and children with other menstrual disorders. Patients 10 years of age and older with non-transfusion-dependent thalassemia syndrome with chronic iron overload require chelation therapy when deferoxamine is contraindicated or inadequate. 2.2. Contraindications Defothal 125 Defothal 125 is contraindicated in the following cases:
Patients with hypersensitivity to the active substance or to any of the excipients of the drug Defothal. Defothal 125 should not be co-administered with other chelation therapies because the safety of these combinations has not been studied. Patients with estimated creatinine clearance < 60 ml/min, estimated GFR less than 40 mL/min/1.73 m2, high-risk myelodysplastic syndrome, platelet count less than 50 x 109/ L.
3. Dosage and how to use Defothal 125
Depending on the user, the dose of Defothal 125 is as follows:
* Adults:
Use the drug with the starting dose: 20mg/kg/day/time. Use the drug with a maintenance dose: 20-40mg/kg/day. Use the drug with the maximum dose: 40mg/kg/day. For adults with thalassemia anemia, use the drug with a starting dose of 10mg/kg orally once a day.
* Children over 2 years old:
Use the drug with the starting dose: 20mg/kg/day/time. Use the drug with a maintenance dose: 20-40mg/kg/day. Use the drug with the maximum dose: 40mg/kg/day. Particularly for children with thalassemia anemia, the starting dose is 10mg/kg orally once a day.
Defothal 125 has not been studied in patients with renal impairment and no dose adjustment is required in the elderly. Particularly for patients with moderate hepatic impairment (Child-Pugh Class B), the dose of Defothal 125 should be reduced significantly, then gradually increased up to the 50% limit.
Defothal is taken orally and the patient should take it once a day, preferably at the same time, on an empty stomach or after a light meal.
4. Defothal 125 . side effects
During the use of Defothal 125, users may experience some of the following side effects:
Common side effects: Headache, diarrhea or constipation, vomiting, abdominal pain, bloating, indigestion , increased transaminases, increased serum creatinine, rash, pruritus. Uncommon side effects: Dizziness, cataracts, laryngeal pain, gastrointestinal bleeding, gastric ulcer, duodenal ulcer, hepatitis, gallstones, pigmentation disorders, renal tubular disorders. Rare side effects: Drug reactions with eosinophilia and systemic symptoms, optic neuritis, esophagitis, fatigue, edema. Side effects of unknown frequency: Thrombocytopenia, anemia, leukopenia, anaphylactic reactions and angioedema, metabolic acidosis, gastrointestinal perforation, acute pancreatitis, liver failure. Some cases had sleep disturbances, hypersensitivity vasculitis, urticaria, erythema multiforme, alopecia, toxic epidermal necrolysis, acute renal failure, tubular interstitial nephritis, nephrolithiasis, tubular necrosis kidney.
5. Drug interactions
Concomitant use of Defothal 125 with strong UGT inducers such as Rifampicin, Carbamazepine, Phenytoin, Phenobarbital, Ritonavir will reduce the effectiveness of Defothal. Defothal reduces the effectiveness of the drugs Cyclosporin, Simvastatin, hormonal contraceptives, Bepridil, Ergotamine) when used concurrently. Concomitant use of Defothal with drugs containing acetylsalicylic acid in high doses, corticosteroids or oral bisphosphonates increases the risk of gastrointestinal toxicity. Concomitant administration of Defothal with anticoagulants may also increase the risk of gastrointestinal bleeding. To avoid interactions with Defothal 125, patients need to inform their doctor about the drugs, vitamins, and dietary supplements they are currently using.
7. Be careful when using Defothal 125
When using Defothal 125, the patient should be monitored for proteinuria, blood count; hearing and vision tests prior to treatment and annually. Change the dose or temporarily discontinue defothal if severe or persistent elevations in serum transaminases are observed. Defothal should not be used in high-risk patients (e.g. myelodysplastic syndromes) or in elderly patients because the drug increases the risk of undesirable effects of Defothal. Monitor for symptoms of peptic ulcer for evaluation and additional treatment if serious gastrointestinal adverse effects of Defohal are suspected. The basic information about Defothal 125 in the above article is for reference only. Because this is a prescription drug, patients should not use it on their own, but need to contact a specialist directly to get an appropriate prescription to ensure safety for health.