This is an automatically translated article.
Post by Master, Doctor Mai Vien Phuong - Gastrointestinal Endoscopy - Department of Medical Examination & Internal Medicine - Vinmec Central Park International General Hospital.
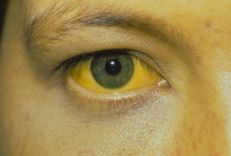
Alcoholic liver disease is initially characterized by fatty liver, which can then develop into alcoholic hepatitis, cirrhosis and cirrhosis. Alcohol can cause damage to many target organs, especially the brain, intestines, and liver. It should be noted that drinking alcohol can alter the structure of the gut microbiota before liver disease occurs.
1. Overview
As an essential mediator of the gut-liver junction, bile acids regulate host-specific metabolic pathways, modulating inflammatory responses through farnesoid-activated receptors and binding bile acid receptors with G protein 1. Several clinical trials have demonstrated the signaling effect of bile acids in the setting of liver disease. The authors hypothesize the existence of the gut microbiota-bile acids-hepatic triangle and explore potential therapeutic strategies for liver diseases targeting this triangle. Bile acids are involved in immune and inflammatory responses in the liver via receptor 1 on the G cell membrane, the G protein-coupled bile acid receptor and the intracellular farnesoid-X receptor. The interaction between the gut microbiota and bile acids can control host immune homeostasis as well as inhibit liver inflammation.
2. Bile acids regulate host immune homeostasis and inflammatory response
Many studies in recent decades have shown that the function of bile acids goes beyond that of "digestive surfactants". At the host level, there is a clear relationship between bile acid signaling and innate immunity in the liver and gut. In other words, bile acids are the cornerstone of the immune axis between the liver and the gut microbiota. As a mediator in the gut-hepatic axis, bile acids can modulate inflammatory responses, host metabolism, and innate immunity, which are effective therapeutic targets in the context of various liver diseases. This is shown in the following table:
| Tên nghiên cứu | Tài liệu Tham khảo | Mục tiêu | Kết quả |
| Các chất chuyển hóa axit mật của vi sinh vật điều chỉnh RORγ ruột + điều hòa cân bằng nội môi của tế bào T | Song và cộng sự, 2020 | Thụ thể hạt nhân BA | Việc khôi phục hệ thống BA trong ruột có thể làm tăng số lượng tế bào RORγ + Treg ở ruột kết và cải thiện tính nhạy cảm của vật chủ đối với bệnh viêm đại tràng. |
| Các chất chuyển hóa axit mật kiểm soát sự phân hóa tế bào Th17 và Treg | Hang và cộng sự , 2019 | Các dẫn xuất của LCA, 3-oxoLCA và isoalloLCA | Sử dụng 3-oxoLCA và isoalloLCA cho chuột làm giảm sự biệt hóa tế bào Th17 và tăng sự biệt hóa tế bào Treg, tương ứng, trong lớp đệm ruột |
| Axit mật kiểm soát chứng viêm và rối loạn chuyển hóa thông qua việc ức chế sự viêm nhiễm NLRP3. | Guo và cộng sự, 2016 | Thụ thể màng TGR5 | Axit mật ức chế sự hoạt hóa của thể viêm NLRP3 thông qua trục TGR5-cAMP-PKA. |
| Chuyển hóa axit mật qua trung gian vi sinh vật đường ruột điều chỉnh ung thư gan thông qua các tế bào | NKT Ma và cộng sự, 2018 | Tế bào CXCR6 + NKT gan | Microbiome sử dụng axit mật như một chất truyền tin để tích tụ các tế bào NKT, có kiểu hình hoạt hóa và ức chế sự phát triển của khối u gan. |
| Hoạt hóa TGR5 ức chế xơ vữa động mạch bằng cách giảm viêm đại thực bào và tải lipid | Pols và cộng sự , 2011 | Kích hoạt TGR5 trong đại thực bào bởi axit 6a-ethyl-23 (S) -methylcholic (6-EMCA, INT-777), một BA bán tổng hợp, ức chế sản xuất cytokine tiền viêm, một hiệu ứng qua trung gian của tín hiệu cAMP do TGR5 gây ra và ức chế NF-κB sau đó |
Abbreviation: PWD: Natural T cell; BA: Bile acids; LCA: lithocholic acid; TGR5: G-protein associated with bile acid receptor 1; Treg: Adjust T; PKA: Protein kinase A; RORγ: RAR-associated monoreading receptor gamma;
However, most contemporary studies on bile acids are based on transgenic mouse models, while the immune system, bile acid metabolism and gut microbiota of mice are very different from those of humans.
3. Alcoholic and bile acid liver disease
Alcoholic liver disease is initially characterized by fatty liver, which can then develop into alcoholic hepatitis, cirrhosis, and cirrhosis. Alcohol can cause damage to many target organs, especially the brain, intestines, and liver. It should be noted that drinking alcohol can alter the structure of the gut microbiota before liver disease occurs. Continued alcohol intake in patients with alcoholic cirrhosis may exacerbate intestinal microbiota disturbances, reduce the detection of intestinal bacteria in stool, and worsen the function of the duodenal and duodenal mucosa. Colon .Chronic alcohol exposure is associated with gut microbiota disturbances in preclinical and human intestinal models as well as the pathogenesis of ALD. Previous studies have shown that the gut microbiota is altered in patients with alcoholic cirrhosis, characterized by an increase in endotoxin-producing bacteria and a decrease in gut bacteria. Changes in the composition of the gut microbiome can alter brain function, and alcohol abuse can also affect the gut-brain axis. This can aggravate alcoholism and cause emotional disturbances that, ultimately, accelerate the development of hepatic encephalopathy.
Compared with non-disease and non-drinkers, patients with alcoholic cirrhosis had higher endotoxin levels and worse gut microbiota disturbances even after alcohol cessation. This indicates that alcohol-induced damage to the gut microbiota continues even after stopping drinking, and that damage also extends to the gut-brain axis, leading to cognitive impairment.
Bệnh gan do rượu ban đầu được đặc trưng bởi gan nhiễm mỡ, sau đó có thể phát triển thành viêm gan do rượu, xơ hóa gan và xơ gan.
FXR can induce the expression of antimicrobial molecules in intestinal epithelial cells to prevent alcohol-induced damage to intestinal tight junctions and avoid loss of intestinal barrier integrity. Gastric administration of FXR agonist fexaramine caused a decrease in serum alanine aminotransferase, hepatic IL-1B, and tumor necrosis factor protein levels. This is because FXR can increase small heterodimer partner proteins to inhibit intestinal inflammation and protect the integrity of the intestinal mucosal barrier. The above findings indicate that FXR agonists can negatively regulate bile acid synthesis and reduce serum bile acid concentrations, which may reduce alcoholic steatosis and hepatitis. Antibiotic use in chronic alcoholics has been found to reduce alcoholic liver disease. This is because antibiotics can kill gut bacteria, reduce bile acid levels, and inhibit the hydrolysis of bile acids. However, it can only reduce the toxicity of deoxycholic acid to hepatocytes and stabilize intestinal barrier function.
In summary, alcohol-related changes in the gut microbiota may ultimately alter bile acid composition. Interventions targeting the bile acid-FXR-FGF15 signaling pathway to regulate CYP7A1 synthesis and lipid metabolism can reduce the occurrence of ALD in mice. As a signaling molecule, bile acids can modulate the complex interactions between alcoholic liver disease that the gut microbiota mediates through the gut-liver-brain axis. Therefore, FGF19 and fexaramine are good approaches for the treatment of ALD.
References:
Shao JW, Ge TT, Chen SZ, Wang G, Yang Q, Huang CH, Xu LC, Chen Z. Role of bile acids in liver diseases mediated by the gut microbiome. World J Gastroenterol 2021; 27(22): 3010-3021 [DOI: 10.3748/wjg.v27.i22.3010]