This is an automatically translated article.
Alendronic acid is an active bisphosphonate used for the prevention of osteoporosis. This active ingredient is in Ortigan. So what does Ortigan do?
1. What is Ortigan?
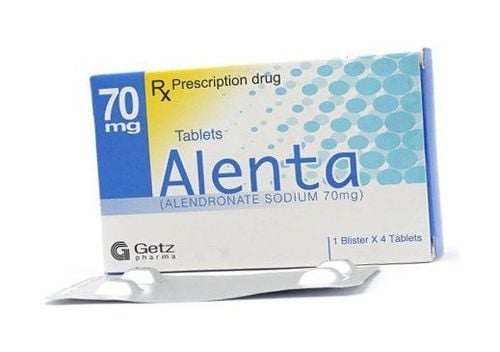
Ortigan medicine has the main active ingredient Sodium Alendronate, equivalent to 70mg Alendronic acid. Ortigan is manufactured in the form of uncoated tablets and packed in a box of 1 blister x 4 tablets manufactured by Cure Medicines (I) Pvt Ltd (India). This product is registered for circulation in Vietnam by Yeva Therapeutics Pvt., Ltd with registration number VN-12785-11.
2. What does Ortigan do?
The active ingredient Alendronic acid in Ortigan is a bisphosphonate that binds mainly to bone resorption sites, especially osteoclasts, to inhibit the activity of osteoclasts without directly affecting bone formation. Because the process of bone resorption and bone formation always go hand in hand, so even though the bone formation process is reduced, the process of bone resorption is inhibited, which also leads to an increase in bone mass. When exposed to Ortigan, normal bone repels Alendronate into the matrix and is not pharmacologically active.3. Indications and contraindications of Ortigan
Ortigan is indicated for the treatment of osteoporosis in postmenopausal women for the prevention of fractures, including fractures of the hip and spine (compression fracture).
Ortigan is contraindicated in the following cases:
Patients with structural abnormalities of the esophagus such as narrowing or loss of elasticity leading to delayed esophageal emptying; The patient's inability to stand or sit upright for at least 30 minutes; Sensitivity or allergy to Alendronic Acid or any of the ingredients in Ortigan products; Patients with hypocalcemia.
4. Dosage, how to use Ortigan
4.1. How to use Ortigan Ortigan used orally, patients must take the drug at least 30 minutes before eating with clean water. Other drinks (including mineral water), food and some medications can reduce the absorption of Ortigan; In order to quickly bring Ortigan to the stomach to reduce the risk of irritation or adverse events occurring locally and in the esophagus, patients should note the following issues: Use Ortigan with a full glass of water upon waking up. during the day; Do not chew or let Ortigan tablets dissolve in the mouth to avoid mouth and throat ulcers; The patient must not lie down for at least 30 minutes after taking Ortigan and until after eating the first meal of the day; Do not take Ortigan before going to bed or while lying in bed when you first wake up during the day; Patients need to supplement calcium and vitamin D during the use of Ortigan if the need from the diet is not enough. 4.2. Dosage of the drug Ortigan Recommended dose: 1 Ortigan 70mg pill, taken only once a week (7 days); No dose adjustment of Ortigan is required for the elderly or patients with mild to moderate renal impairment; Ortigan is not recommended for patients with more severe renal impairment (creatinine clearance less than 35 mL/min) due to lack of clinical experience; The optimal duration of Ortigan's dosing has not been established. All patients receiving bisphosphonate therapy should be continued and reevaluated periodically. 4.3. Ortigan Overdosage and Treatment No special information is available on the treatment of Ortigan overdose ; Overdose of Alendronic acid can lead to hypocalcemia and hypophosphataemia, the appearance of gastrointestinal side effects such as dysfunction, heartburn, esophagitis or gastric ulcer; Treatment: Give the patient milk or an antacid to combine with Alendronate. Note that due to the risk of esophageal irritation, patients receiving an overdose of Ortigan should not induce vomiting and should always remain in a fully upright position. 4.4. Missed dose of Ortigan and how to handle it The doctor should instruct the patient that if he or she forgets to take a dose of Ortigan 70mg, it must be taken in the morning as soon as it is discovered; Absolutely do not take 2 Ortigan tablets on the same day, but return to take a single pill a week on the same day of the week as the original schedule.
5. Ortigan side effects
In clinical studies Ortigan was generally well tolerated. In long-term studies, the adverse effects of Ortigan were generally mild and did not require discontinuation of treatment.
The following adverse reactions have been reported post-marketing of Ortigan:
Hypersensitivity reactions including urticaria and, rarely, angioedema; Like other bisphosphonates, alendronate may cause transient symptoms such as myalgia, malaise, weakness and fever, especially at the beginning of treatment; Hypocalcaemia (rare); Peripheral edema (rare); Nausea, vomiting, esophagitis/slippery/ulcer, which can rarely cause esophageal stricture or perforation and oropharyngeal ulceration; Stomach or duodenal ulcers, sometimes severe and with complications; Localized osteonecrosis of the jaw, often associated with tooth extraction and/or local infection (including osteomyelitis) and long-lasting; Bone pain, joint pain and/or muscle pain, rarely severe; Loss of mobility; Joint swelling, upper femur fracture due to slight impact; Dizziness; Taste disorder; Skin rash (occasionally with increased photosensitivity); Itching, hair loss; Rarely serious skin reactions such as Stevens Johnson syndrome and toxic epidermal necrolysis; Rarely, choroiditis, scleritis or inflammation of the sclera; Transient low serum calcium and phosphate, without clinical symptoms.
6. Ortigan's Drug Interactions
If taken at the same time, calcium supplements, drugs that reduce stomach acidity (antacids) and other oral medications may affect the absorption of Ortigan. Therefore, patients need to wait at least 30 minutes after taking Ortigan to start taking other drugs.
Use of hormone replacement therapy (Estrogen plus Progestin) plus Ortigan was evaluated in 2 clinical trials of 5 or 5 years in postmenopausal women with osteoporosis for higher bone mass results. , while reducing bone turnover more than treating each therapy alone. At the same time, these studies show that the safety and tolerability of the combination is similar to that of the individual drugs.
Ortigan was used in studies of osteoporosis in postmenopausal men and women with a range of commonly prescribed drugs with no clinically relevant adverse reactions.
Non-steroidal anti-inflammatory drugs (NSAIDs) can cause stomach and intestinal irritation, so caution should be exercised when combining with Ortigan.
7. Be careful when using Ortigan
Like other bisphosphonates, Ortigan may cause local irritation of the upper gastrointestinal mucosa. There have been reports of esophageal adverse events, such as inflammation, ulceration and erosions of the esophagus, however, narrowing or perforation has been rare in patients treated with Ortigan. Some of them are severe enough to require hospitalization. Therefore, the physician should be cautious of symptoms suggestive of esophageal reactions, thereby instruct the patient to stop taking Ortigan and notify the doctor immediately, such symptoms include difficulty swallowing, swallowing pain, pain after the breastbone, new or more severe heartburn. The risk of serious adverse reactions to the esophagus is increased if the patient takes Ortigan incorrectly and/or continues to take it after symptoms of esophageal irritation. Therefore, the doctor needs to provide enough information for the patient to understand the use and dosage of Ortigan. At the same time, the doctor should warn the patient that if the instructions for taking Ortigan are not followed, the risk of side effects in the esophagus may increase. Ortigan can cause irritation of the upper gastrointestinal mucosa and has the potential to make the condition worse, so doctors should be cautious when prescribing Ortigan to patients with upper gastrointestinal problems. such as difficulty swallowing, esophageal disease (including Barrett's esophagus), peptic ulcer disease or inflammation. In order for Alendronate to reach the stomach quickly and thereby reduce the risk of esophageal irritation, the doctor should instruct the patient to take Ortigan with a full glass of water and not to lie down for at least 30 minutes after taking it. until after eating for the first time of the day. There have been a few (rare) reports of localized osteonecrosis of the jaw (ONJ), often associated with tooth extraction and/or local infection (including osteomyelitis) with prolonged resolution, when taking oral bisphosphonates (including Ortigan). The majority of cases of osteonecrosis of the jaw due to Bisphosphonate are cancer patients treated intravenously. Known risk factors for ONJ include cancer, combination therapy (Ortigan plus chemotherapy, radiation or corticosteroids), poor oral hygiene, and comorbidities (such as periodontal disease and/or disease). or dental disease, anemia, blood clotting disorders, infections) and smoking. Patients with focal osteonecrosis of the jaw (ONJ) should be cared for and monitored by a dental practitioner, and Ortigan should be discontinued according to the outcome of the individual risk/benefit assessment. body. Oral procedures can make this effect worse. Therefore, before performing dental procedures (e.g. extractions, dental implants), the treating physician and/or dentist should make a clinical assessment and prescribe a course of action, including the use of Ortigan is based on a risk/benefit assessment for each individual patient. Patients taking bisphosphonates (such as Ortigan) may experience bone pain, joint pain, and/or muscle pain. However, these symptoms are rarely severe, and/or disabling. The duration of these symptoms ranged from 1 day to many months after starting treatment with Ortigan, and most improved when the drug was stopped. There have been a number of reports of mild stress fractures below the trochanter and superior head of the femur in patients receiving bisphosphonates (including Ortigan), including fractures (a type of fracture). incomplete bone) not due to trauma. Some patients sometimes have the forewarning symptom of pain in the fracture area and the appearance of a fracture for a time before the actual fracture occurs. The physician should carefully evaluate the patient with suspected fracture, including assessment of the cause and risk factors (such as vitamin D deficiency or malabsorption, corticosteroid use, history of previous fractures, degeneration, etc.) joint or lower extremity fracture, excessive or heavy exercise, diabetes or metabolic disease, chronic alcoholism), then the patient should receive appropriate specialist trauma care. Discontinuation of Ortigan in patients with fractures may be considered, depending on the individual patient's risk/benefit assessment. Ortigan is not recommended for patients with creatinine clearance less than 35 mL/min. Before treatment with Ortigan, patients should be considered and evaluated for all causes of osteoporosis other than estrogen deficiency and aging. Hypocalcaemia must be corrected prior to initiation of Ortigan therapy. Other disorders of mineral metabolism (such as vitamin D deficiency) also need to be actively managed. Due to the positive effect of Alendronate in increasing bone mineralization, Ortigan may cause a transient (asymptomatic) decrease in serum calcium and phosphorus concentrations. Alendronate has not been studied in children, so Ortigan is not indicated for this population. The effects of Ortigan on the ability to drive and use machines have not been studied. However, some undesirable effects have been reported with Ortigan, which may affect the ability to drive and use machines. Alendronate has not been studied for use in pregnant and lactating women, so Ortigan should not be used in these subjects.
Please dial HOTLINE for more information or register for an appointment HERE. Download MyVinmec app to make appointments faster and to manage your bookings easily.