This is an automatically translated article.
The article was written by Doctor Hoang Quoc Chinh - Stem Cell Research Institute and Gene Technology - Vinmec Times City International General HospitalTo date, the US Food and Drug Administration (FDA) has approved eight immune checkpoint inhibitor therapies for the treatment of several cancers including lung, colorectal, and liver cancers. , breasts, and head and neck. This list will continue to grow in the near future as clinical trials of immune checkpoint inhibitors continue to roll out worldwide.
1. General overview of immune checkpoint inhibitor therapy
Checkpoint inhibitor therapies are therapies that release immunosuppression by removing signals that inhibit T cell activation/activity. When activated by a “foreign” antigen (antigen) and a costimunatory signal, T cells divide to produce millions of antigen-specific T cells. These antigen-specific T cells secrete cytokines, interferons, and participate in active killing to eliminate pathogens.The key to an effective immune system is the ability to distinguish pathogens from the body's normal cells, and attack only "foreign" cells. To be able to distinguish "foreign" cells from normal cells in the body, T cells must pass through two selection processes: central tolerance and peripheral tolerance. Central tolerance occurs in the thymus when the immature T cells divide to produce a large number of T cells. The gene segments that code for the T cell receptors (TCR) of the these T cells are also recombined to produce TCRs that can recognize all the antigens that T cells can encounter. T cells that interact strongly with major histocompatibility complexes (MHCs) carrying self-peptides are eliminated to avoid autoimmune reactions (T cells attack normal cells). normally of the body). Central tolerance also removes T cells that do not interact with MHC and retains only T cells with weak MHC interactions. T cells selected after central toleration will be recirculated to participate in peripheral tolerance.
Peripheral tolerance involves the development of a series of immune control mechanisms i.e. inhibition of T-cell activation and/or activity, to ensure that autoimmune processes do not occur. CTLA-4 (cytotoxic T lymphocyte-associated antigen 4) and PD-1 (program death 1) are the two most studied immune control pathways. Knowledge of the immunosuppressive mechanisms of CTLA-4 and PD-1, as well as preclinical evidence for antitumor activity of CTLA-4 and PD-1 inhibitors, laid the scientific foundation for development of immune checkpoint inhibitors in the treatment of cancer.
To date, the US Food and Drug Administration (FDA) has approved 8 immune checkpoint inhibitor therapies for the treatment of several cancers including lung, colorectal, and liver cancers. , breast, and head and neck (details in Table 1). This list will continue to grow in the near future as clinical trials of immune checkpoint inhibitors continue to roll out worldwide.
This article will present an overview of the immunosuppressive mechanism, the immune release mechanism of immune checkpoint inhibitors in cancer treatment, and biomarkers to help select patients for therapy. This method aims to provide the basic knowledge about immunosuppressive therapy.
Table 1: List of FDA-approved immune checkpoint inhibitors
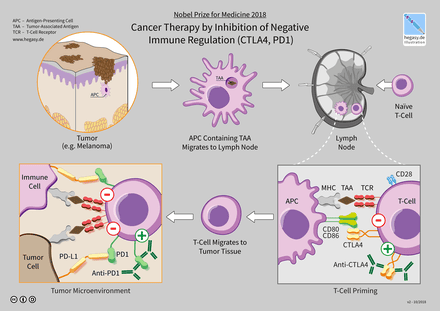
2. Immune checkpoint inhibition via CTLA-4 and PD-1 . receptors
2.1. Activation of T cells
Tế bào T.
With Helper T Cells, the CD28 receptor is the key molecule of co-activation signaling. When CD28 on helper T cells interacts with B7.1 (CD80) or B7.2 (CD86) receptor on the surface of APC cells, the T cell begins to divide to produce millions of specific T cells. antigenic signaling and production of agents necessary for the elimination of pathogens. Only when T-cell TCR and CD28 interact with antigen-MHC and B7 complexes on the surface of APC cells, respectively, is T-cell activation activated. Unlike helper T cells, cytotoxic T cells do not depend much on the CD28 molecule but on CD70 and 4-1BB receptors (CD137) for their activation. Once activated, the T cell will express the genes necessary for its survival and leave the lymph node to enter the tumor. Here, effector T cells recognize cancer cells and destroy them by secreting cytokines, interferons, and enzymes that stimulate cell apoptosis. Besides promoting T-cell division and differentiation, T-cell activation also stimulates the expression of immunosuppressive pathways to ensure that T-cells attack only agents. cause disease rather than harming the body's normal cells.
2.2. Immune control via the CTLA-4 . receptor
The first immunosuppressive signal described was the CTLA-4 receptor. CTLA-4 is structurally homologous to CD28, but has a higher affinity for B7 on the APC surface than CD28, so it competes with the co-activation signal by blocking the B7-CD28 interaction, thereby inhibiting transactivation. In addition, several studies have shown that the interaction of CTLA-4 with B7 induces a signal that inhibits T-cell activation through competition for ligands. With co-activation receptor (CD28) and activating inhibitory signal, CTLA-4 plays an important role in controlling T-cell activation. The immunoregulatory function of CTLA-4 is well established. reinforced by the fact that mice without the ctla-4 gene will overproduce lymphocytes and do not live beyond 3-4 weeks of age.CTLA-4 increased expression level as soon as TCR recognized the foreign MHC-antigen complex and reached maximum levels two or three days after T cells were activated. In addition to increasing the expression level of CTLA-4, "activated" T cells also "transport" CTLA-4 from the cytoplasm to the immune synapse of T cells. The concentration of CTLA-4 in the immunoreceptor region is directly proportional to the degree of T-cell activation (the more activated the T-cell, the more CTLA-4 is present in the immunoreceptor site). When the number of CTLA-4:B7 complexes in the immunoreceptor region far exceeds the number of CD28:B7 complexes, T-cell activation is completely inhibited, thereby eliminating the cellular immune response. T.
CTLA-4 has also been shown to be required for the immunosuppressive function of regulatory T cells. “Control” T cells have an important role in peripheral immune tolerance and their function is impaired in the absence of CTLA-4. One of the immunosuppressive mechanisms of “control” T cells is to reduce the expression of the B7 molecule on the surface of APC cells, thereby reducing coactivation signaling through the CD28 receptor of the cell. T cells.
2.3. PD-1 . receptor-mediated immune control
Kiểm soát miễn dịch qua thụ thể PD-1.
The interaction of the PD-L ligand with the PD-1 receptor transmits an immunosuppressive signal to the T cells, thus leading to an "exhausted T cell" state. Like CTLA-4, PD-1 also inhibits T-cell activity through reduced coactivation of the CD28 receptor, but PD-1 does not compete with CD28 for interaction with the B7 ligand, but rather inhibits CD28 receptor activity. CTLA-4 controls T-cell activation in the lymphatic system, but PD-1 controls activated T-cell activity in peripheral tissues. The role of the PD-1 receptor in immune control is evident through the autoimmunity of animals that do not have the Pdcd-1 gene (the gene that encodes for PD-1) and who can carry it. mutation of this gene. In addition, the PD-1/PD-L1 pathway is also thought to play a role in controlling T-cell infiltration into the tumor, thereby preventing direct T-cell contact. activated and cancer cells.
3. Cancer treatment mechanism of CTLA-4 and PD-1/PD-L1 . inhibitor therapy
Insights into the immunosuppressive mechanisms of the CTLA-4 and PD-1 pathways, as well as the results of preclinical studies on antitumor resistance and increased survival in animal models for cancer of CTLA-4 and PD-1 inhibitors laid the scientific foundation for the development of immune checkpoint inhibitor therapy. Several monoclonal antibodies that inhibit the activity of the CTLA-4 and PD-1 pathways have been approved by the U.S. Drug Administration for the treatment of several types of cancer (Table 1), and being tested on other cancers. Although the mechanism of action of CTLA-4 and PD-1/PD-L1 inhibitors has not been fully elucidated, the current model suggests that CTLA-4 inhibitors act on T cells in The activated phase takes place in the lymph nodes while the PD-1/PD-L1 inhibitor acts on activated T cells in the tumor.
The primary mechanism of immunosuppression of CTLA-4 inhibitors is through blocking the interaction of the CTLA-4 receptor with the B7 ligand, thereby allowing T cells to continue to be activated through the receptor. line B7/CD28. The spatial structure of a mixture of CTLA-4 with ipilimumab, a CTLA-4-binding antibody, shows that ipilimumab directly interacts with CTLA-4 at the site where B7 binds to CTLA-4. This implies that ipilimumab locked the interaction site of CTLA-4 with B7, thereby preventing the inhibition of T-cell activation [10]. Recent evidence suggests that CTLA-4 receptor inhibitors increase the number of antigen-specific cytotoxic T cells of cancer cells, not just T cells in general. CTLA-4 receptor inhibitors also decrease the inhibitory function of “control” T cells, thereby increasing T cell antitumor resistance. In short, CTLA-4 receptor inhibitors increases T-cell antitumor resistance through immunosuppressive “elimination”.
PD-1/PD-L1 pathway inhibitors have the ability to eliminate tumors through reactivation of toxic T cells, thereby increasing the activity and percentage of toxic T cells. Blocking the PD-1 receptor releases immunosuppression of “exhausted” T cells, thereby allowing “inactivated” T cells to be reactivated and increasing the efficiency of the immune system. immunohistochemistry despite the presence of PD-L in the tumor. Clinical trial evidence suggests that PD-1/PD-L inhibitors are most effective against tumors that already have activated toxic T cells, but are inhibited by the PD-1/PD-L1 pathway. . However, an immune response has also been observed in some tumors that do not express the PD-1 ligand PD-L1 or PD-L2. The antitumor activity of PD-1/PD-L1 inhibitors also requires the presence of helper T cells. Although PD-1/PD-L1 inhibitors have received much attention and research, their anticancer mechanisms have not been completely elucidated.
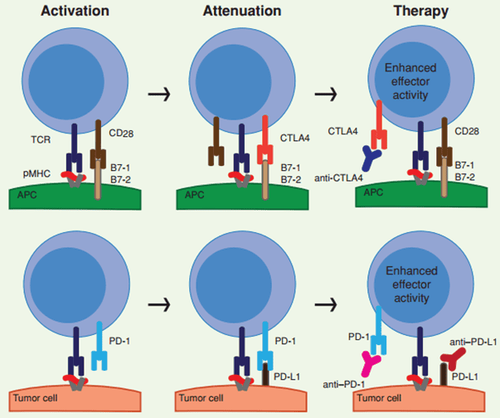
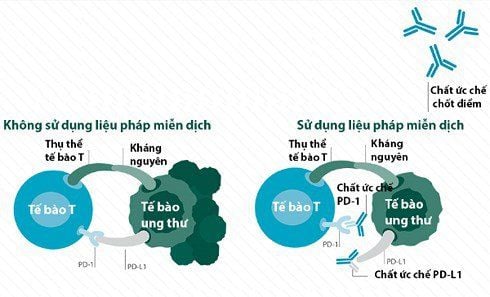
Cơ chế giải phóng ức chế miễn dịch của liệu pháp ức chế chốt kiểm soát miễn dịch.
(A) T cells, after being activated by the pMHC:TCR complex and B7:CD28 (activated T cells), express CTLA-4. CTLA-4 competes for B7 with CD28 and inhibits T cells (suppresses T cells). The antibody CTLA-4 (anti-CTLA4) interacts with CTLA-4, thereby releasing immunosuppression (inhibitory release).
(B) PD-1 is expressed in activated T cells (activated T cells). Tumor cell PD-L1 interacts with PD-1 to inactivate T cells (T-cell suppression). Antibodies to PD-1 (anti-PD-1) or PD-L1 (anti-PD-L1) interact with PD-1 or PD-L1 respectively, thereby reactivating "inactivated" T cells (dissolving). inhibitory release).
4. Indications for immunosuppressive therapy
CTLA-4 and PD-1/PD-L1 inhibitors outperform traditional therapies in improving the quality and survival of many cancer patients, but the number of patients who respond treatment remains below 50% for single therapies while combination therapies are costly. Therefore, selecting patients who are likely to respond to this therapy will play an important role in maximizing the benefits of this therapy. Currently, the expression level of PD-L1 and the state of MSI (microstalite instability) in the tumor are licensed markers for the selection of suitable patients for PD-1/PD-L1 inhibitor therapy. Clinical trials to select markers capable of predicting treatment response to immune checkpoint inhibitor therapy include tumor burden mutation and gene markers. gene signatures) are being implemented.
Please dial HOTLINE for more information or register for an appointment HERE. Download MyVinmec app to make appointments faster and to manage your bookings easily.