This is an automatically translated article.
Degarelix belongs to a group of anti-cancer drugs commonly used to treat prostate cancer in men. The drug works by reducing the amount of testosterone the body makes to stop or slow the growth of cancer cells. However, Degarelix also has some side effects that patients need to be aware of when using.1. What is Degarelix?
Degarelix is a synthetic peptide derivative that binds to gonadotropin-releasing hormone (GnRH) receptors in the pituitary gland and prevents interaction with GnRH. It is this antagonism that reduces the hormone LH and follicle-stimulating hormone (FSH) which ultimately causes the suppression of testosterone from the testes. The reduction of testosterone is important in the treatment of prostate cancer in men. Therefore, Degarelix is often indicated for the control of advanced prostate cancer.2. Dosage of the drug Degarelix
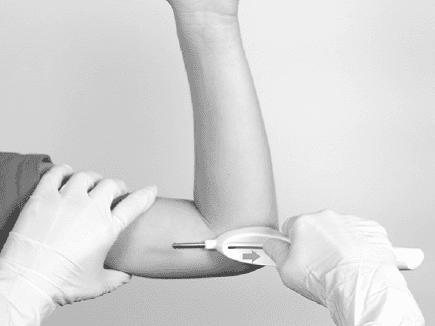
Degarelix is for subcutaneous injection only, not for intravenous use and intramuscular injection is not recommended because there are no specific studies. Degarelix must be mixed before use, usually Degarelix will be injected under the skin of the abdomen and the injection site should be changed periodically, should be injected into areas where the patient is not under pressure such as avoiding areas near the waist or waistband. and not close to the ribs.
Reference dose of Degarelix is as follows:
Starting dose: 240mg divided into 2 subcutaneous injections, each time 120mg Maintenance dose - monthly use: 80mg once subcutaneously The first maintenance dose should be administered after 1 month starting dose In cases where the patient's clinical response is suboptimal, it is advisable to determine whether serum testosterone levels are still adequately suppressed as Degarelix does not cause a sudden increase in testosterone levels. Therefore, additional antiandrogen therapy is not required to avoid a sudden increase in the initiation of therapy. Note that no dose adjustment is necessary in elderly patients or in patients with mild or moderate renal or hepatic impairment. Use with caution in patients with severe hepatic or renal impairment There are no relevant indications for the use of Degarelix in women, children and adolescents.
3. Degarelix side effects
The most commonly reported adverse reactions during treatment with Degarelix are due to the possible pharmacological effect of testosterone suppression. These include hot flashes and weight gain or injection site reactions. Transient chills, fever or flu-like illness have also been reported to occur several hours after administration.
Side effects at the injection site were mainly pain and erythema, swelling, stiffness, and vesicles, mainly at the starting dose, while with the maintenance dose these reactions were less common. Some other side effects that may occur when using Degarelix include:
Blood and lymphatic system disorders Hypersensitivity reactions Metabolic and nutritional disorders Weight gain, hypertension, diabetes mellitus, increased cholesterol, changes Blood calcium changes Insomnia Dizziness, headache, mental decline, decreased sensation Blurred vision Arrhythmias (atrial fibrillation), palpitations, QT prolongation Hypertension, vasomotor reactions Difficulty breathing Constipation, vomiting, abdominal pain, abdominal discomfort, dry mouth Increased bilirubin, elevated alkaline phosphatase Urticaria, skin papillomas, hair loss, pruritus, erythema Osteoporosis, bone loss, arthralgia, muscle weakness, contractions Dysuria, urgency, dysuria, nocturia, renal failure Testicular pain, breast tenderness, pelvic pain, genital irritation Discomfort, peripheral edema
4. Drug interactions with Erythromycin
Some common drug interactions with Degarelix include:
Do not use Degarelix concurrently with drugs that prolong the QT interval or drugs that can cause torsades de pointes such as class I antiarrhythmic drugs (eg, quinidine, disopyramide). as androgen deprivation therapy itself may prolong the QT interval Degarelix is not a substrate for the human CYP450 system and has not to any extent induce or inhibit CYP1A2, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1 in vitro, so it is unlikely that there is a drug-drug pharmacokinetic interaction in the metabolism involving these isoenzymes. Please note the above important information so that the use of drugs in patients is effective, safe and minimizes side effects. If you have any problems, you should talk to your doctor directly for specific advice.
Please dial HOTLINE for more information or register for an appointment HERE. Download MyVinmec app to make appointments faster and to manage your bookings easily.